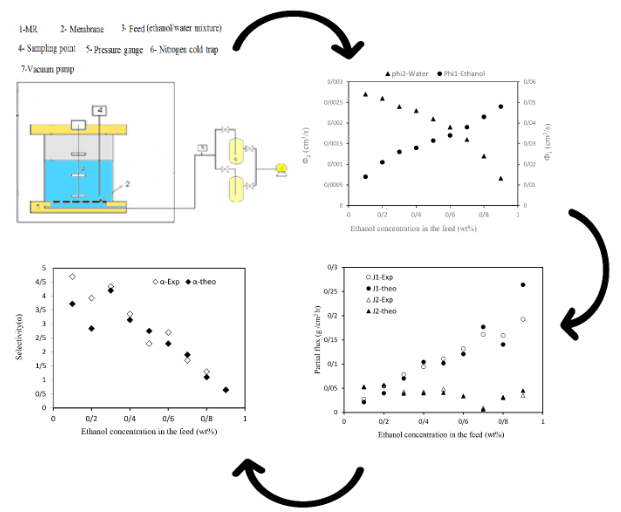
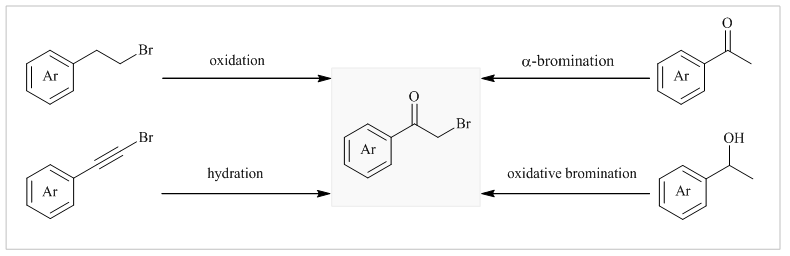
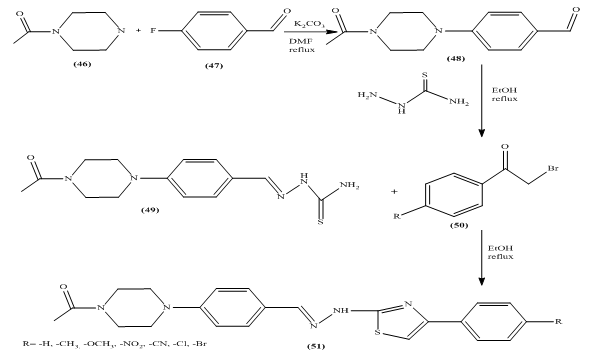
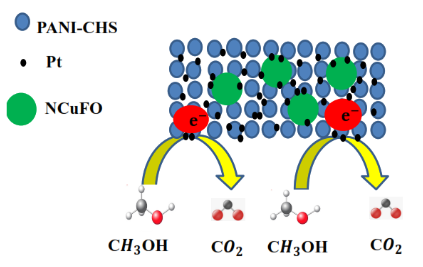
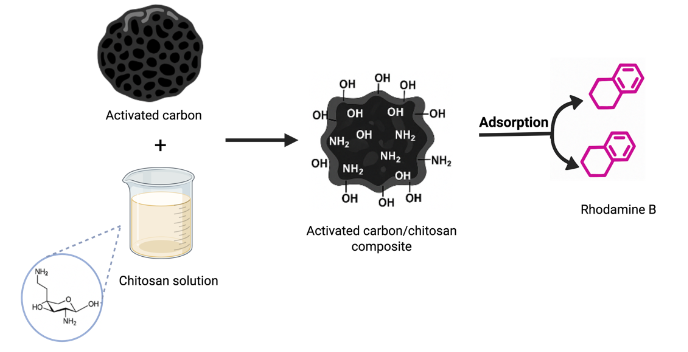
Evaluation of polydimethylsiloxane membrane performance for ethanol/water mixture separation: Mathematical modeling
Pages 659-672
https://doi.org/10.22034/crl.2025.502132.1532
Marzieh Faghih, Mehri Esfahanian
Abstract Since bioethanol is produced as a green fuel in an aqueous solution, improving the process of separating of ethanol/ water mixture is crucial due to the presence of an azeotropic point in the distillation process. Therefore, ethanol separation processes, conducted concurrently with its production in bioreactors, with an emphasis on membrane-based processes and focusing on pervaporation, are preferred due to their lower cost and energy requirements for ethanol separation. In this study, the design and construction of a pervaporation unit were first carried out. Then, the ethanol-water separation process was modeled using the Solution-Diffusion model and the Flory-Huggins theory in pervaporation. The dense polydimethylsiloxane (PDMS) membrane was used due to its superior selectivity for ethanol. The results showed that ethanol could be separated from water at any feed composition without any azeotropic point. Furthermore, by calculating the diffusion coefficients of ethanol and water from the model and ultimately determining the partial ethanol and water fluxes, it was observed that the calculated flux values from the model closely matched the experimental flux values for each component. Additionally, increasing the ethanol concentration in the feed led to an increase in ethanol’s diffusion coefficient, a decrease in water’s diffusion coefficient, an increase in total flux, and ultimately a reduction in the separation factor, which were in good agreement with the experimental selectivity values obtained from the experiments. This indicates the validity of using the Flory-Huggins model for predicting ethanol separation from water in the pervaporation process.