Determination of Fe(III) ion with a novel, highly efficient immobilized nitrosa R-salt in a polymer matrix
Pages 448-459
https://doi.org/10.22034/crl.2025.496212.1505
M. B. Kholboyeva, Z. A. Smanova, U. A. Madatov, S. B. Rakhimov, B.T Orzikulov, Abror Karim ugli Nomozov, M.R Uralova
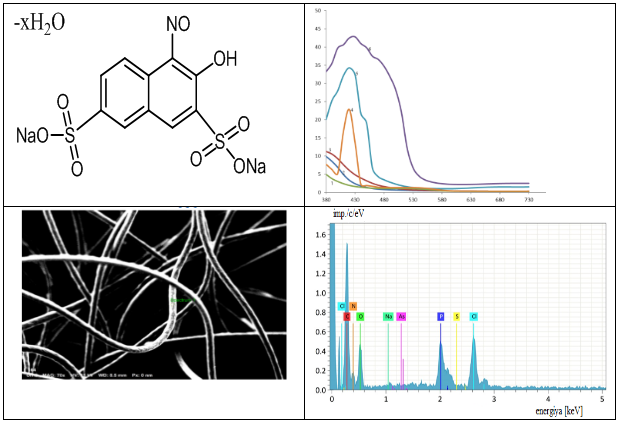
Abstract In this paper, nitrosa R salt immobilized on PPF matrix was used as an analytical reagent for the determination of Fe(III) ion. The advantage of the proposed method over existing methods is the local synthesis of the carrier sorbent used and the higher analytical parameters for the determination of Fe(III) ion (Sr=0.05, Cmin=0.2μg/l). In order to assess the accuracy of the method, the t-criterion and F-criterion were compared with the tabular data (Texperiment=1.09; ttable=2.83, Fexperiment=2.52; Ftable=4.47). When determining Fe(III) using nitrosa R salt used for immobilization in acetate buffer solution at pH=5.5, the obtained complex gave a maximum analytical signal at a wavelength of 720 nm. The linear binding obeys the Beer-Lambert-Beer law in the range of 0.2-2 μg/ml. Under selected conditions, common coexisting ions usually do not interfere with the determination of iron ions in water samples. The proposed method is simple, economical, rapid, sensitive, accurate and highly selective, and can be used to determine the iron content in various water samples without prior treatment..