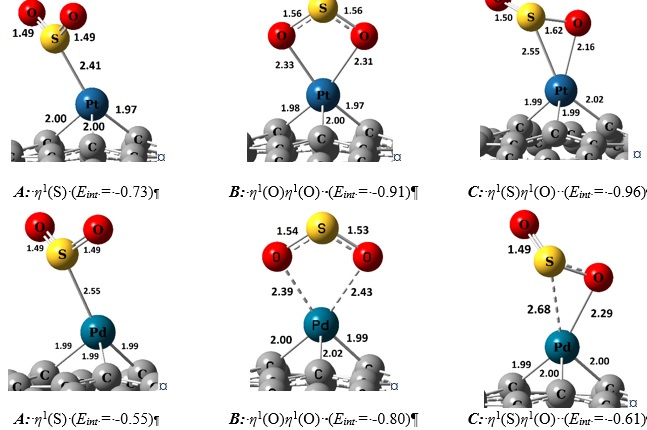
Enhanced adsorption performance and efficiency of graphitic GaN monolayers through functionalizing with transition metal adatoms (Co, Cu and Ni): A DFT study
Pages 912-925
https://doi.org/10.22034/crl.2024.467458.1375
Zahraa Saba Ghnim, Ayat Husein Adthab, Morug Salih Mahdi, Aseel Salah Mansoor, Usama Kadem Radi, Nasr Saadoun Abd, Rebaz Obaid Kareem
Abstract In this work, we have accomplished first principles calculations to scan the geometrical and electronic behaviors of GaN substrates functionalized with transition metal (Co, Cu and Ni) adatoms. The adsorption capacity of the Ni-adsorbed GaN monolayers was also evaluated to take advantage of applying these improved nanosystems in gas sensor devices. Among these transition metal adatoms, the highest adsorption energy and geometrical stability belong to the Ni adsorption on the GaN monolayer. All the transition metals prefer the nitrogen site of GaN monolayer for adsorption. Besides, all the transition metal adsorbed GaN monolayers unveil semiconducting activities due to the band gap rise around the Fermi level. The charge densities were comprehensive between the transition metals and nitrogen atoms of GaN system, indicating the chemical reaction between them. Subsequently, the adsorption features of CO, NO and NO2 on the surface of pristine and Ni-adsorbed GaN were surveyed in detail. The results indicated that the gas molecules are sturdily chemisorbed on the Ni-adsorbed GaN. Therefore, Ni functionalization expressively enhanced the sensitivity of GaN monolayer for gas trapping. Our results highlighted that the Ni-adsorbed GaN monolayers will help to encourage scientists to develop improved 2D GaN-based sensor systems in the future.