Kerim Avaz Kuliyev, Nailya Allahverdi Verdizadə, K.R. Alieva, Şafa Ağa Mamedova Mamedova
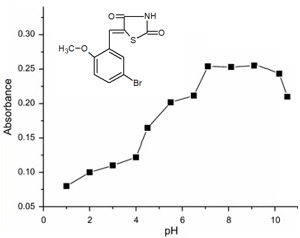
Abstract A simple and selective spectrophotometric method was developed for the determination of nickel(II) using 5-(2-bromo-5-methoxybenzylidene)-thiazolidine-2,4-dione (BMBT) as a chromogenic reagent forming a yellow brown complex. The possibility of using BMBT for photometric determination of nickel(II) has been studied. The nickel(II) complex is formed in the pH range 7.1-8.9. To achieve the best formation and extraction of the complex, a BMBT concentration of 5×10-4 mol/l is recommended. The study tested several solvents, including water (as the base medium), methyl alcohol, ethyl alcohol, chloroform, hexane, dichloromethane, and acetonitrile. Among these, methanol demonstrated the most favorable outcomes, making it the best-suited solvent for the experiment.
The maximum analytical signal for the complexation of nickel with BMBT is observed at 482 nm. The shift of the absorption peak to the long wavelength side - the bathochromic shift in this case is 146 nm. The molar absorption coefficient is 1.75×104. The ratio of components in the complex is Ni: BMBT =1:2. It has been established that Ni2+ is a complexing form of nickel and displaces one hydrogen atom from one BMBT molecule. The structure of the complex was studied by thermogravimetry and IR spectroscopy.
The study showed that when BMBT is used as a chromogenic reagent, colored complexes are also formed by various metal ions, including Fe(III), Cu(II), Mo(VI), Se(IV), Pt(II), Mn(II), Cd(II), Zn(II), Pb(II) and Pd(II). The accuracy of nickel determination in the presence of these metal ions increases significantly in the presence of masking agents or by changing the pH of the solution.Obedience to the basic law of light absorption is observed at a nicke concentration of 0.3-13 μg / ml. The interfering influence of extraneous ions is eliminated with the help of masking substances, changing the pH of the medium or using extraction. Based on the results of a spectrophotometric study of the nickel(II) complex with BMBT, methods have been developed for the determination of nickel in various objects.